Synthesis of an imidazolinium chloride via cyclisation of a diamine
SyntheticPage 31
DOI:
Submitted: July 16, 2001, published: July 16, 2001
Authors
Lisa Titcomb (lisa.titcomb@gmail.com)
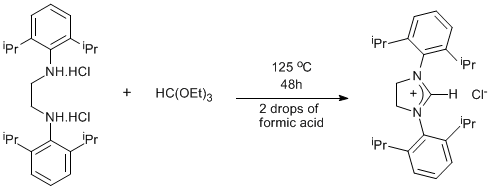
Chemicals
N,N’-Bis-(2,6-diisopropylphenylamine)ethane dihydrochloride (see Page 30)
Triethyl orthoformate (Aldrich, distilled from sieves onto sieves)
Formic acid (Aldrich)
Triethyl orthoformate (Aldrich, distilled from sieves onto sieves)
Formic acid (Aldrich)
Procedure
N,N’-Bis-(2,6-diisopropylphenylamine)ethane dihydrochloride (37.33g, 0.082 mol) was placed in a round bottom flask with a stirbar. Triethyl orthoformate (463 mL) and formic acid (4 drops) were added. The flask was fitted with a condenser and the mixture was heated at reflux for 48h open to the air. After cooling to ambient temperature a white crystalline solid formed. The solid was collected by filtration and placed in a round bottomed flask. The excess triethyl orthoformate was removed by heating the solid at 70oC under vacuum (1x10-2mbar) for 8h to yield 28g of the product (80% yield).
Author Comments
The reaction mixture reaches reflux at a temperature of 125oC (cf triethyl orthoformate bp=146oC). If the reaction is carried out in a sealed ampoule heated at 146oC the yield drops to 30%. The excess triethyl orthoformate is easily removed by heating under vacuum rather than repeated recrystallizations. The Cl- counterion can be exchanged for BF4- by dissolving the imidazolinium salt (1mmol) in water (20mL) with ammonium tetrafuoroborate (1mmol). A white solid precipitates which when dried under vacuum yields the BF4 salt (95% yield).
Data
1H NMR (D6-DMSO): 1.14 (d, 12H, iPrCH3, 3J(HH) = 6.9 Hz), 1.30 (d, 12H, iPrCH3, 3J(HH) = 6.9 Hz), 3.03 (sept, 4H, iPrCH, 3J(HH) = 6.9 Hz), 4.49 (s, 4H, CH2), 7.3-7.5 (m, 6H, aryl-CH), 9.52 (s, 1H, NCHN).
Lead Reference
Arduengo, A. J.; Krafczyk, R.; Schmuter, R. Tetrahedron, 1999, 55, 14523.
Other References
For next stage in carbene prep see Page 34
Keywords
cyclisation, electrophilic, imidazolinium salt, imidazolium salt