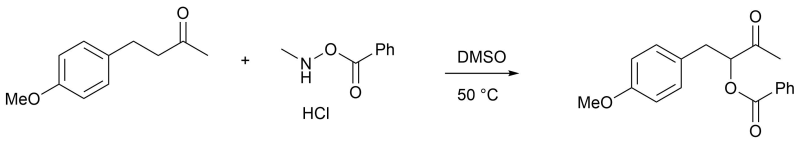
α-Oxidation of Ketone
SyntheticPage 912
Submitted: December 16, 2019, published: January 14, 2020
Authors
Yoichiro Hirose (yoichiro.hirose@tcichemicals.com)
Yuko Konishi (yuko.konishi@tcichemicals.com)
Yuta Yamamoto (yuta.yamamoto@tcichemicals.com)
Chemicals
o-Benzoyl-N-methylhydroxylamine Hydrochloride (TCI)
Procedure
To a solution of 4-(4-methoxyphenyl)-2-butanone (0.679 mL, 4.0 mmol) in DMSO (10 mL) was added o-benzoyl-N-methylhydroxylamine hydrochloride (884 mg, 4.5 mmol) and the mixture was stirred at 50°C for 12 hours. Deionized water (100 mL) was added to the reaction mixture. After being stirred for 10 minutes, the mixture was extracted with ethyl acetate (100 mL) and the separated organic layer was washed with 2M hydrochloric acid (100 mL), saturated aqueous sodium bicarbonate solution (100 mL) and brine (100 mL), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by column chromatography (3:97–30:70 ethyl acetate / hexane on SiO2), giving slightly yellow oil (0.90 g, 75%).
Author Comments
Data
1H NMR (400 MHz, CDCl3); δ 2.09 (s, 3H), 3.14 (ddd, 2H, J = 14.8, 5.2, 2.4 Hz), 3.76 (s, 3H), 5.37 (dd, 1H, J = 5.2, 2.8 Hz), 6.82 (td, 2H, J = 8.8, 2.6 Hz), 7.17 (td, 2H, J = 8.8, 2.6 Hz), 7.44 (tt, 2H, J = 7.6, 1.6 Hz), 7.57 (tt, 1H, J = 7.2, 2.0 Hz), 8.02 (dd, 2H, J = 7.2, 1.6 Hz)
Lead Reference
Supplementary Information
Keywords
ketones, oxidation