Free-radical hydrostannylation and in situ iododestannylation
SyntheticPage 84
DOI:
Submitted: August 16, 2001, published: August 17, 2001
Authors
Stephen Caddick (s.caddick@sussex.ac.uk)
A contribution from
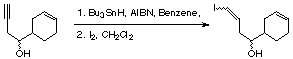
Chemicals
Substrate,
Bu3SnH (1 equiv),
AIBN (0.1 equiv),
benzene (1 - 2 ml / mmol),
iodine (1.2 equiv),
CH2Cl2 (1 - 2 ml / mmol) .
Bu3SnH (1 equiv),
AIBN (0.1 equiv),
benzene (1 - 2 ml / mmol),
iodine (1.2 equiv),
CH2Cl2 (1 - 2 ml / mmol) .
Procedure
TBTH (0.9 ml, 3.3 mmoles) was added to a stirred solution of the alcohol (498 mg, 3.3 mmoles) and AIBN (57 mg, 0.35 mmoles) in benzene (5 ml) and heated for one hour. The reaction was then cooled to room temperature (reaction monitored by tlc). The solvent removed in vacuo and the crude product dissolved in CH2Cl2 (5 ml) and iodine added at room temperature.The reaction mixture was stirred until complete by tlc (few hours to overnight). The reaction mixture was diluted with CH2Cl2 (50 ml) and the organic layer washed with saturated sodium thiosulphate solution (3 x 20 ml), brine (3 x 20 ml) and dried over sodium sulfate.Purification by silica-gel chromatography (petrol: ether, 20:1, 15:1, 10:1, then 5:1). Partial seperation of isomers achieved (373mg, 40%) then mixture (83 mg, 9%) then 35 mg, 6%).
Author Comments
This is very easy and can be quick because the hydrostannylation can be done very concentrated and the iodination is almost certainly instantaneous as you can see the dissipation of the iodine colour on addition to the stannane. Purification is not too difficult although the use of a KF work-up might be advisable in some cases. The transformation of a vinyl stannane to a vinyl iodide proceeds with retention of configuration.
Although I have only done this reaction on a few hundred mg to perhaps 2g I am sure it will scale up well.
Data
z-isomer only
1H NMR
1-2.4, m, 10H; 3.6, m, 1H; 5.7, s, 2H; 6.4, s, 2H;
Lead Reference
For a general reference see: Pereyre, M; Quintard, J. P.; Rahm, A. Tin in Organic Synthesis, Butterworths, 1987
Keywords
Comments
If there is excess Tributyltinhydride how it will be quenched?
It will come along with compound during colum.
By ravigullapudi@yahoo.com on June 22, 2008