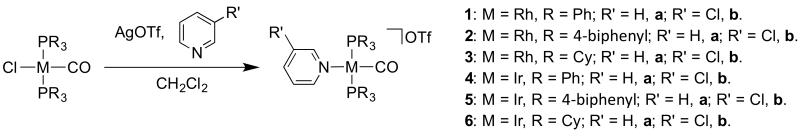
Halide abstraction and addition of pyridine at rhodium and iridium centres
SyntheticPage 809
DOI:
Submitted: August 10, 2016, published: August 16, 2016
Authors
Adrian Chaplin (a.b.chaplin@warwick.ac.uk)
Richard Knighton (r.c.knighton@warwick.ac.uk)
Rory Finn (r.finn@warwick.ac.uk)
Thibault Troadec (t.troadec@warwick.ac.uk)
A contribution from
Chemicals
Dichloromethane
Pentane
Pyridine
3-Chloropyridine
Silver(I) trifluoromethansulfonate
[Rh(PPh3)2(CO)Cl] (ref 1)
[Rh(P(4-biphenyl)3)2(CO)Cl] [[808]]
[Rh(PCy3)2(CO)Cl] (ref 2)
[Ir(PPh3)2(CO)Cl] (ref 3)
[Ir(P(4-biphenyl)3)2(CO)Cl] [[808]]
[Ir(PCy3)2(CO)Cl] (ref 4)
Procedure
A J. Young’s flask protected from light was charged with [M(PR3)2(CO)Cl] (ca. 100 mg, 1.0 eq.) and AgOTf (1.1 eq.). A solution of pyridine or 3-chloropyridine (5 eq.) in CH2Cl2 (5 mL) was added and the resulting mixture was stirred at room temperature for 1 hour. The solution was filtered to remove precipitated AgCl salts and the solvent removed in vacuo. The resulting yellow solid was washed with pentane (2 × 3 mL) and then recrystallized by slow diffusion of pentane into a CH2Cl2 solution of the compound.
Author Comments
All manipulations were performed under an atmosphere of nitrogen, using Schlenk and glove box techniques. Glassware was oven dried at 150ºC overnight and flamed under vacuum prior to use. Anhydrous CH2Cl2 and pentane were purchased and handled strictly under an inert atmosphere. CD2Cl2 was dried over CaH2, vacuum distilled, and freeze-pump-thaw degassed three times before being placed under argon.
Data
[Rh(PPh3)2(CO)(py)]OTf (1a)
Yield = 55%.1H NMR (500 MHz, CD2Cl2) δ 7.58 (d, JHH = 5.4, 2H, py), 7.45-7.56 (m, 18H, Ph), 7.37-7.44 (m, 12H, Ph), 7.36 (d, JHH = 8.4, 1H, py), 6.67 (t, JHH = 6.8, 2H, py). 13C{1H} NMR (126 MHz, CD2Cl2) δ 150.8, 138.7, 133.7, 131.3, 130.0 (t, JPC = 24), 129.7, 126.3. The CO resonance was not unambiguously identified. 31P{1H} NMR (122 MHz, CD2Cl2) δ 31.5 (d, JRhP = 128). ESI-MS (180 °C, 4 kV): positive ion: 655.0840 m/z, [M-py]+ (calcd. 655.0821 m/z). IR (CH2Cl2, cm-1): ν(CO) 2010. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498244.
[Rh(PPh3)2(CO)(3-Clpy)]OTf (1b)
Yield = 38%. 1H NMR (500 MHz, CD2Cl2) δ 7.75 (d, JHH = 7.8, 1H, py), 7.48-7.61 (m, 18H, Ph), 7.43 (dd, JHH = 8.5, 6.8, 12H, Ph), 7.37-7.29 (m, 2H, py), 6.76 (dd, JHH = 8.2, 5.5, 1H, py). 13C{1H} NMR (126 MHz, CD2Cl2) δ 190.1 (dt, JRhC = 69, JPC = 16), 149.7, 148.9, 138.3, 134.3 (t, JPC= 7), 132.1, 130.3 (t, JPC = 24), 129.7 (t, JPC = 5) 126.7. 31P{1H} NMR (122 MHz, CD2Cl2) δ 31.8 (d, JRhP = 127). ESI-MS (180 °C, 4 kV): positive ion: 655.0833 m/z, [M-Clpy]+ (calcd. 655.0821 m/z). IR (CH2Cl2, cm-1): ν(CO) 2013. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498245.
[Rh(P(4-biphenyl)3)2(CO)(py)]OTf (2a)
Yield = 79%. 1H NMR (500 MHz, CD2Cl2) δ 7.78 (d, JHH = 5.3, 2H, py), 7.69-7.75 (m, 24H, Ar), 7.63-7.65 (m, 12H, Ar), 7.47-7.52 (m, 12H, Ar), 7.41-7.45 (m, 6H, Ar), 7.37 (t, JHH = 7.7, 1H, py), 6.73 (t, 2H, JHH = 6.8, py). 13C{1H} NMR (126 MHz, CD2Cl2) δ 151.2, 144.7, 139.8, 138.5, 134.9 (t, JPC = 7), 129.7, 129.3 (t, JPC = 25), 129.0, 128.2 (t, JPC = 5), 127.7, 126.4. The CO resonance was not unambiguously identified. 31P{1H} NMR (202 MHz, CD2Cl2) δ 43.5 (d, JRhP = 128). ESI-MS (180 °C, 4 kV): positive ion: 1111.2698 m/z, [M-py]+ (calcd. 1111.2699 m/z). IR (CH2Cl2, cm-1): ν(CO) 2009. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498246.
[Rh(P(4-biphenyl)3)2(CO)(3-Clpy)]OTf (2b)
Yield = 83%. 1H NMR (500 MHz, CD2Cl2) δ 8.06 (d, JHH = 5.5, 1H, py), 7.72-7.81 (m, 24H, Ar), 7.64-7.67 (m, 12H, Ar), 7.48-7.52 (m, 13H, Ar+py), 7.41-7.45 (m, 6H, Ar), 7.31 (d, JHH = 8.3, 1H, py), 6.88 (dd, JHH = 8.3, 5.5, 1H, py). 13C{1H} NMR (176 MHz, CD2Cl2) δ 190.3 (dt, JRhC= 69, JPC = 17), 150.1, 149.3, 144.9, 139.9, 138.0, 134.9 (t, JPC = 7), 134.1, 129.6, 129.0, 129.0 (t, JPC = 24), 128.3 (t, JPC = 5), 127.7, 127.0. 31P{1H} NMR (202 MHz, CD2Cl2) δ 30.4 (d, JRhP = 127). ESI-MS (180 °C, 4 kV): positive ion: 1111.2685 m/z, [M-py]+ (calcd. 1111.2699 m/z). IR (CH2Cl2, cm-1): ν(CO) 1995. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498247.
[Rh(PCy3)2(CO)(py)]OTf (3a)
Yield = 42%. 1H NMR (500 MHz, CD2Cl2) δ 8.47 (d, 2H, JHH = 7.3, py), 8.01 (t, 1H, JHH = 8.0, py), 7.60-7.57 (m, 2H, py), 1.78-1.91 (m, 24H, Cy), 1.61-1.76 (m, 12H, Cy), 1.46-1.61 (m, 12H, Cy), 1.25 (qt, JPH = 13.1, JHH = 3.6, 6H, Cy), 1.04 (qt, JPH = 13.0, JHH = 3.3, 12H, Cy). 13C{1H} NMR (126 MHz, CD2Cl2) δ 192.6 (dt, JRhC = 69, JPC = 16), 152.1, 140.1, 126.9, 35.4 (t, JPC = 10), 31.0, 28.1 (t, JPC = 5), 26.9. 31P{1H} NMR (122 MHz, CD2Cl2) δ 33.1 (d, JRhP = 118). ESI-MS (180 °C, 4 kV): positive ion: 691.3652 m/z, [M-py]+ (calcd. 691.3638 m/z). IR (CH2Cl2, cm-1): ν(CO) 1983. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498248.
[Rh(PCy3)2(CO)(3-Clpy)]OTf (3b)
Yield = 19%. 1H NMR (500 MHz, CD2Cl2) δ 8.51 (br, 1H, py), 8.40 (d, JHH = 5.4, 1H, py), 8.04 (dd, JHH = 8.4, JHH = 1.5, 1H, py), 7.70 (dd, JHH = 8.4, JHH = 5.5, 1H, py), 1.80-1.93 (m, 24H, Cy), 1.75-1.64 (m, 12H, Cy), 1.46-1.62 (m, 12H, Cy), 1.31-1.21 (m, 6H, Cy), 1.13-0.98 (m, 12H, Cy). 13C{1H} NMR (126 MHz, CD2Cl2) δ 192.3 (dt, JRhC = 70, JPC = 16), 150.7, 150.2, 140.0, 134.7, 128.0, 35.5 (t, JPC = 10), 31.1 (d, JPC = 16) 28.1 (m), 26.9. 31P{1H} NMR (122 MHz, CD2Cl2) δ 33.1 (d, JRhP = 117). ESI-MS (180 °C, 4 kV): positive ion: 691.3649 m/z, [M-py]+ (calcd. 691.3638 m/z). IR (CH2Cl2, cm-1): ν(CO) 1986. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498249.
[Ir(PPh3)2(CO)(py)]OTf (4a)
Yield = 85%. 1H NMR (500 MHz, CD2Cl2) δ 7.53-7.60 (m, 14H, Ph + py), 7.49 (t, JHH = 7.4, 6H, Ph), 7.39-7.44 (m, 12H, Ph), 7.32 (t, JHH = 7.8, 1H, py), 6.67-6.73 (m, 2H, py). 13C{1H} NMR (126 MHz, CD2Cl2) δ 174.9 (t, JPC = 11), 150.9, 139.3, 134.6 (t, JPC = 6), 132.1, 130.1 (t, JPC = 28), 129.6 (t, JPC = 5), 126.8. 31P{1H} NMR (122 MHz, CD2Cl2) δ 26.9. ESI-MS (180 °C, 4 kV): positive ion: 824.1821 m/z, [M]+ (calcd. 824.1820 m/z). IR (CH2Cl2, cm-1): ν(CO) 2000. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498250.
[Ir(PPh3)2(CO)(3-Clpy)]OTf (4b)
Yield = 72%. 1H NMR (500 MHz, CD2Cl2) δ 7.72 (d, JHH = 5.6, 1H, py), 7.56-7.61 (m, 12H, Ph), 7.49-7.53 (m, 6H, Ph), 7.41-7.46 (m, 12H, Ph), 7.33 (d, JHH = 8.4, 1H, py), 7.27 (d, JHH = 2.3, 1H, py), 6.81 (dd, JHH = 8.3, 5.6, 1H, py). 13C{1H} NMR (126 MHz, CD2Cl2) δ 174.4 (t, JPC = 11), 149.9, 149.0, 138.9, 134.5 (t, JPC = 6), 132.3, 129.7, (t, JPC = 28), 129.6 (t, JPC = 5), 127.3. 31P{1H} NMR (122 MHz, CD2Cl2) δ 27.2. ESI-MS (180 °C, 4 kV): positive ion: 745.1404 m/z, [M-py]+ (calcd. 745.1398 m/z). IR (CH2Cl2, cm-1): ν(CO) 2003. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498251.
[Ir(P(4-biphenyl)3)2(CO)(py)]OTf (5a)
Yield = 91%. 1H NMR (500 MHz, CD2Cl2) δ 7.75-7.80 (m, 14H, Ar + py), 7.69-7.73 (m, 12H, Ar), 7.62-7.66 (m, 12H, Ar), 7.48-7.52 (m, 12H, Ar), 7.41-7.45 (m, 6H, Ar), 7.34-7.39 (m, 1H, py), 6.75-6.79 (m, 2H, py). 13C{1H} NMR (176 MHz, CD2Cl2) δ 175.1 (t, JPC = 11), 151.3, 144.9, 139.8, 139.0, 135.1 (t, JPC = 6), 129.7, 129.0, 128.8 (t, JPC = 28), 128.2 (t, JPC = 5.8), 127.7, 126.9. 31P{1H} NMR (202 MHz, CD2Cl2) δ 39.0. ESI-MS (180 °C, 4 kV): positive ion: 1242.3528 m/z, [M-py+MeCN]+ (calcd. 1242.3546 m/z). IR (CH2Cl2, cm-1): ν(CO) 1999. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498252.
[Ir(P(4-biphenyl)3)2(CO)(3-Clpy)]OTf (5b)
Yield = 86%. 1H NMR (500 MHz, CD2Cl2) δ 8.01 (d, JHH = 5.5, 1H, py), 7.77-7.83 (m, 12H, Ar), 7.72-7.75 (m, 12H, Ar), 7.62-7.66 (m, 12H, Ar), 7.48-7.52 (m, 12H, Ar), 7.41-7.45 (m, 7H, Ar + py), 7.30 (d, JHH = 8.5, 1H, py), 6.93 (dd, JHH = 8.5, 5.5, 1H, py). 13C{1H} NMR (176 MHz, CD2Cl2) δ 174.7 (t, JPC = 12), 150.2, 149.5, 145.1, 139.8, 138.5, 135.1 (t, JPC = 6), 134.7, 129.7, 129.1, 128.5 (t, JPC = 27), 128.2 (t, JPC = 5), 127.8, 127.6. 31P{1H} NMR (202 MHz, CD2Cl2) δ 39.3. ESI-MS (180 °C, 4 kV): positive ion: 1242.3550 m/z, [M-py+CH3CN]+ (calcd. 1242.3546 m/z). IR (CH2Cl2, cm-1): ν(CO) 2002.
[Ir(PCy3)2(CO)(py)]OTf (6a)
Yield = 83%. 1H NMR (400 MHz, CD2Cl2) δ 8.47 (d, 2H, JHH = 5.2, py), 8.05 (t, JHH = 7.6, 1H, py), 7.55-7.72 (m, 2H, py), 1.66-1.95 (m, 36H, Cy), 1.51-1.66 (m, 12H, Cy), 1.18-1.31 (m, Cy), 0.97-1.10 (m, 12H, Cy). 13C{1H} NMR (101 MHz, CD2Cl2) δ 176.6 (t, JPC = 11), 152.1, 140.7, 127.5, 35.5 (t, JPC = 13), 30.9, 28.1 (t, JPC = 5), 26.9. 31P{1H} NMR (122 MHz, CD2Cl2) δ 28.4. ESI-MS (180 °C, 4 kV): positive ion: 781.4221 m/z, [M-py]+ (calcd. 781.4215 m/z). IR (CH2Cl2, cm-1): ν(CO) 1975. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498253.
[Ir(PCy3)2(CO)(3-Clpy)]OTf (6b)
Yield = 38%. 1H NMR (500 MHz, CD2Cl2) δ 8.51 (d, JHH = 2.3, 1H, py), 8.42 (d, JHH = 5.5, 1H, py), 8.08 (d, JHH = 8.4, 1H, py), 7.79 (dd, JHH = 8.4, 5.5, 1H, py), 1.66-2.03 (m, 36H, Cy), 1.49-1.67 (m, 12H, Cy), 1.20-1.31 (m, 6H, Cy), 0.96-1.15 (m, 12H, Cy). 13C{1H} NMR (126 MHz, CD2Cl2) δ 176.1 (t, JPC = 11), 150.8, 150.3, 140.5, 135.2, 128.6, 35.6 (t, JPC = 13), 30.9 (d, JPC = 21), 28.1 (t, JPC = 5), 26.9. 31P{1H} NMR (122 MHz, CD2Cl2) δ 28.5. ESI-MS (180 °C, 4 kV): positive ion: 781.4203 m/z, [M-py]+ (calcd. 781.4215 m/z). IR (CH2Cl2, cm-1): ν(CO) 1979. The solid-state structure of this compound has been deposited with the Cambridge Crystallographic Data Centre under CCDC 1498254.Lead Reference
Some of these complexes have been reported previously, e.g. 1a and 1b, C. Woods , C. Daffron, Inorg. Chim. Acta 1985, 101, 13-16 http://dx.doi.org/10.1016/S0020-1693(00)85619-4; 3a, J. Am. Chem. Soc. 1981, 103, 3411–3422 http://dx.doi.org/10.1021/ja00402a027
Other References
| 1. M. C. Baird, C. J. Nyman, G. Wilkinson, J. Chem. Soc., A 1968, 348–351. http://dx.doi.org/10.1039/j19680000348 |
||||
| 2. S. Otto, A. Roodt, Inorg. Chim. Acta 2004, 357, 1–10. http://dx.doi.org/10.1016/S0020-1693(03)00436-5 |
||||
| 3. J. P. Collman, C. T. Sears and M. Kubota, Inorg. Synth., 1990, 28, 92–94. http://dx.doi.org/10.1002/9780470132593.ch24 |
||||
| 4. M. J. Burk, R. H. Crabtree, Inorg. Chem. 1986, 25, 931–932. http://dx.doi.org/10.1021/ic00227a010 |
||||
Keywords
carbonyl, iridium, organometallics, phosphine, pyridine, rhodium