Alkylation of 1-indanone
SyntheticPage 696
DOI:
Submitted: September 12, 2013, published: September 22, 2013
Authors
A contribution from
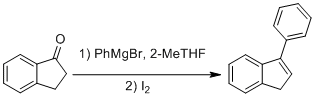
Chemicals
1-Indanone (≥99%, ReagentPlus, Sigma-Aldrich)
Phenylmagnesium Bromide (2.8 M solution in 2-MeTHF, Acros Organics)
2-MeTHF (≥99%, Anhydrous, Sigma-Aldrich)
Iodine (≥99.8%, ACS Reagent, Sigma-Aldrich)
Diethyl Ether (Sigma-Aldrich)
Saturated Ammonium Chloride
10% Aqueous Sodium Thiosulfate
Procedure
The synthesis was performed using standard Schlenk techniques under a nitrogen atmosphere. To a sealed roundbottom flask charged with 1-indanone (2.00 g, 0.0151 mol), anhydrous 2-MeTHF (76 mL) was added. Phenylmagnesium Bromide (5.4 mL, 0.0151 mol, 2.8 M solution in 2-MeTHF) was added dropwise to the reaction mixture at 0 oC. The reaction mixture was allowed to return to room temperature. The reaction mixture was then stirred at 40 oC for 20 h. The reaction mixture was quenched and washed with saturated ammonium chloride (3 x 15 mL) and extracted with diethyl ether (3 x 15 mL). The organic layer was dried over magnesium sulfate, filtered into a roundbottom flask, and further eluted with diethyl ether (10 mL). The solvent was evaporated under reduced pressure. Crude 1-phenyl-indanol was isolated. To the roundbottom flask charged with the crude mixture was added: iodine (383 mg, 1.51 mmol). The neat reaction mixture was stirred at RT for 20 h. TLC was used to monitor the reaction.* The reaction was quenched with aqueous sodium thiosulfate (10% aqueous, 3 x 15 mL) and diethyl ether (3 x 15 mL). The reaction mixture was dried over magnesium sulfate, filtered into a roundbottom flask, and further eluted with diethyl ether (10 mL). The solvent was evaporated under reduced pressure, yielding a tan oil. The product was then purified using flash chromatography (95:5 Hex:EtOAc) to yield a yellow oil, 3-phenyl-indene (1.88 g, 65%).
*An aliquot was removed from the crude reaction mixture and diluted for TLC.
Author Comments
The yield of the 2 g synthesis was determined by isolating the product. This synthesis was attempted at a 3 g scale and the isolated yield was determined to be 53%. This synthesis was also attempted at a 100 mg scale and the isolated yield was determined to be 65%.
A saturated solution of sodium chloride (Brine) was used to remove emulsions during the washing phases.
Data
1H NMR (CDCl3, 500 MHz): δ ppm 3.56 (d, J = 2.3 Hz, 2H), 6.64 (t, J = 2.0 Hz, 1H), 7.27-7.68 (m, 9H)
1H NMR is similar to previously printed spectra. See lead reference.
Lead Reference
Stavber, G.; Zupan, M.; Stavber, S. Tetrahedron Lett., 2006, 47, 8463-8466. http://dx.doi.org/10.1016/j.tetlet.2006.09.154
Keywords
alkenes, alkylation, elimination, ketones, organometallics, solventless