Thermal Ring Contraction of a 1,4-Diazepine
SyntheticPage 574
DOI:
Submitted: November 15, 2012, published: November 15, 2012
Authors
Ramesha Ramakrishna (ramesha63@hotmail.com)
A contribution from
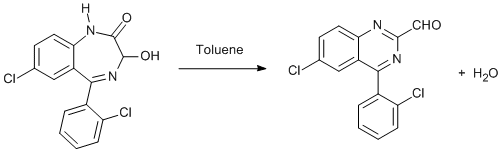
Chemicals
Toluene commercial, 99%
Procedure
Author Comments
2. This is a clean method for the synthesis of 6-Chloro-4-(o-chlorophenyl)-2-quinazoline carboxaldehyde
3. The procedure does not requires any purification. The product is almost insoluble in the reaction medium and therefore it crystalizes out from the reaction medium.
Data
Melting pont : 176-178 oC
1H NMR (CDCl3, 300 MHz): δ δ 10.31 (s, 1H), 8.30 (d, J = 6.7 Hz,1H), 7.99 (dd, J = 6.6,1.6 Hz, 1H), 7.72 (d, J = 1.7 Hz, 1H), 7.52-7.48 (m, 4H)
13C NMR (CDCl3, 75 MHz): δ 191.3, 167.9, 155.3, 149.1, 136.6, 136.1, 134.8, 132.7, 131.7, 131.4, 130.9, 130.2, 127.3, 126.0, 124.8.
FTIR (KBr): 1721, 1385, 823 cm-1;
Lead Reference
Other References
1. Rutgers, J. G.; Shearer, C. M Analytical Profiles of Drug Substance, Vol 9, 1980, Edited by Florey K., Acedemic Press, 1980, 397-426. 2. Nudelman, A, McCaully, R. J, Bell, S. C. J. Pharm. Sci., 1974, 63, 1880-1884.
3. Sternbatch, L. H., Reder, E., Stempel, A., Rachlin, A. I. J. Org.Chem.,1964, 29, 332-336.
Supplementary Information
Keywords
aldehydes, amides, aromatics/arenes, elimination, impurity, Lorazepam, quinazoline aldehyde, thermal, thermal rearrangement