Perchlorination of highly deactivated aromatic compounds
SyntheticPage 483
DOI:
Submitted: February 10, 2011, published: February 11, 2011
Authors
Nenad Maraš (nenad.maras@gmail.com)
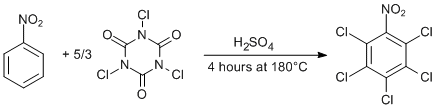
Chemicals
Nitrobenzene (Fluka)
Trichloroisocyanuric acid (Fluka)
H2SO4
Procedure
A mixture of nitrobenzene (1.25 g, 10 mmol) and sulfuric acid (96%, 10 ml) was stirred on an oil bath at room temperature while trichloroisocyanuric acid (5.0 g, 20 mmol) was carefully added. After 15 min of stirring a short air condenser was set on the flask and the temperature of the oil bath slowly increased to 180°C during the period of 2 h. The reaction mixture was left stirring for 4 h at that temperature. The organic layer solidified during cooling and was extracted directly from the sulfuric acid with dichloromethane (40 ml), the extract washed with water (3 × 40 ml), dried over anhydrous Na2CO3 and evaporated in vacuo (Caution: The waste acid is still concentrated and should be carefully quenched and disposed). The resulting 2.58 g of greenish crystals were triturated in hot methanol and filtered when cold to give white crystalline 2,3,4,5,6-pentachloronitrobenzene (1.86 g, 62 %).
Author Comments
The use of trichloroisocyanuric acid in sulfuric acid for electrophilic aromatic chlorination was first described by Rosevear and Wilshire. Perchlorination of some deactivated aromatic substrates like phthalic anhydride and 4-alkylnitrobenzenes with this reagent combination at about 100°C is published in two Japonese patents. Mendonça et al published a thorough study of chlorination with trichloroisocyanuric acid in sulfuric acid.
The crude product of the above reaction contains also some 2,3,4,5-tetrachloronitrobenzene (1H NMR singlet at 7.90 ppm in CDCl3) and 2,3,5,6-tetrachloronitrobenzene (singlet at 7.74 ppm) in the molar ratio of 2 : 1 respectively. Both are effectively removed by trituration in hot methanol. Up-scaling the reaction resulted in somewhat reduced yields, presumably due to the less effective stirring of the biphasic system which forms already at the beginning.Data
mp = 141–143°C (lit. 145–146 °C). IR (KBr) 3500, 1559, 1364, 1333, 1223, 928, 785, 676, 656 cm-1. MS (EI) m/z (%) 295 (M+, 68), 249 (83), 237 (100), 214 (75), 142 (83); the isotopic distribution is consistent with 5 Cl atoms.
Lead Reference
Maraš, N.: Funkcionalizacija alkenov in aromatov s trikloroizocianurno kislino (diploma). University of Ljubljana, Ljubljana, 2006.
Other References
Uzuki, H.; Hashiba, I. JP9067359, 1997.
Koichi, H.; Tadahisa, S. JP9268159, 1997.
Mendonça, G. F.; Magalhães, R. R.; de Mattos, M. C. S.; Esteves, P. M. J. Braz. Chem. Soc., 2005, 16, 695–698.
Keywords
alkyl/alkenyl/aryl halides, aromatics/arenes, electrophilic, substitution