Oxidative substitution of cobalt nitrate
SyntheticPage 296
DOI:
Submitted: August 28, 2008, published: August 28, 2008
Authors
Neetisha Mistry (n.s.mistry@warwick.ac.uk)
A contribution from
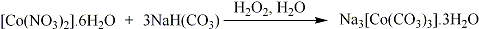
Chemicals
Co(NO3)2.6H2O;
NaH(CO3);
H2O2 (30%)
NaH(CO3);
H2O2 (30%)
Procedure
NaH(CO3) (42.185 g, 0.502 mol) was added to water (50 ml). The solution was stirred and placed in an ice bath. A solution of Co(NO3)2.6H2O (29.118 g, 0.100 mol), water (50 ml), and H2O2 (30%, 10 ml) was added dropwise to the ice cold slurry. On addition of the solution, the reaction mixture turned green. The mixture was kept at 0 °C for a further 1½ hours, whilst being continuously stirred. The green product was filtered, washed with cold water (3 x 10 ml), ethanol (2 x 10 ml), and diethyl ether (2 x 10 ml). The dark green precipitate was left to dry overnight under dynamic vacuum. Yield 31.9 g, 88%
Author Comments
Product was washed thoroughly by stirring the product in the solvents, then filtering at a buchner funnel. Literature references gave similar yields.
Data
IR(cm-1): 3238 (O-H), 1451 (C=O), 1328 (C=O). CHN % found (calculated for C3H6CoNa3O12): C 10.09 (9.95), H 1.29 (1.67)
Lead Reference
A.Damiamoglou et al. A New Reference Material for UV-visible Circular Dichroism Spectroscopy, Chirality, 2008
H.F.Bauer and W.C.Drinkard, J. Am. Chem. Soc., 1960, 82, 5031
Keywords
Comments
We use this to make an EDDS complex (http://www.syntheticpages.org/pages/298) and have found that it should be freshly prepared. Over time stored in air, the carbonate darkens.
By Peter Scott on May 7, 2009