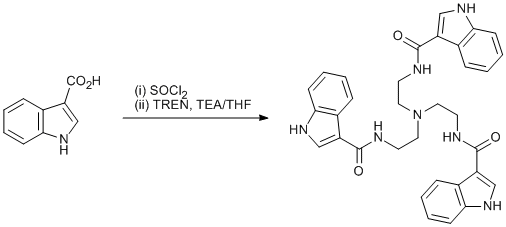
Amination of an indole acyl chloride
SyntheticPage 265
DOI:
Submitted: January 6, 2010, published: November 30, 2007
Authors
Christine R. Whitlock (cwhitlock@georgiasouthern.edu)
Chemicals
indole-3-carboxylic acid,
thionyl chloride,
tris(2-aminoethyl)amine(TREN),
triethylamine,
tetrahydrofuran
thionyl chloride,
tris(2-aminoethyl)amine(TREN),
triethylamine,
tetrahydrofuran
Procedure
Indole-3-carboxylic acid (0.56 g, 3.4 mmol) was dissolved in SOCl2 (6 mL) and stirred at 0°C for 1.5 h. The solution was concentrated in vacuo and to the resulting oil was added TREN (0.62 g, 4.2 mmol) and TEA (0.46 g, 4.6 mmol) in THF (25 mL). After stirring at 0°C for 30 min, the solution was filtered under vacuum to yield 5 as a beige solid which was recrystallized in MeOH.
Author Comments
This reaction is a reliable way to prepare tris(2-[indole-3-amido]ethyl)amine.
Data
Yield 0.28 g (43.0%); mp > 250°C. 1H NMR (250 MHz, DMSO-d6) 7.98 (s, 3H, H2), 7.44 (m, 3H, H1), 7.17 (m, 3H, H4), 7.15 (d, 3H, H7), 7.14 (m, 3H, H5 or H6), 7.12 (m, 3H, H6 or H5), 3.40 (m, 12H, Ha and Hb); MS (M+1)+ = 576 for C33H33N7O3.
Lead Reference
Sears, R. Bryan; Carpenter, Russell A.; Whitlock, Christine R. “A General Synthesis of Tris-Indole Derivatives as Potential Iron Chelators.” Molecules, A Journal of Synthetic Chemistry and Natural Product Chemistry, 2005, 10, 488-491.