3,6-disubstitution of sarcosine anhydride with indoles
SyntheticPage 218
DOI:
Submitted: May 20, 2004, published: May 22, 2004
Authors
Christine R. Whitlock (cwhitlock@georgiasouthern.edu)
A contribution from
Chemicals
sarcosine anhydride (Aldrich)
o-dichlorobenzene (Aldrich)
bromine (Aldrich, 99.5%)
hexanes (Aldrich)
indole (Aldrich)
N,N-dimethylformamide (Aldrich)
methanol (Fisher)
o-dichlorobenzene (Aldrich)
bromine (Aldrich, 99.5%)
hexanes (Aldrich)
indole (Aldrich)
N,N-dimethylformamide (Aldrich)
methanol (Fisher)
Procedure
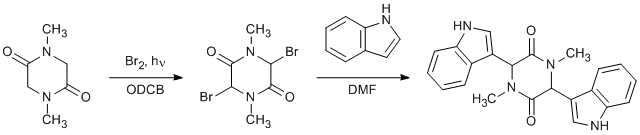
To a solution of sarcosine anhydride (3.75 g, 26.4 mmol) in o-dichloromethane (15 mL), at 150°C, was added dropwise Br2 (3.0 mL, 58.1 mmol) in o-dichlorobenzene (6 mL), under illumination of a sun lamp. The solution was heated for 1 h and then cooled to room temperature. A stream of N2 was bubbled through the reaction mixture for 10 min, and the solution was diluted with hexanes. The resulting beige crystals of 3,6-dibromo-1,4-dimethylpiperazine-2,5-dione were filtered and rinsed with hexanes (7.06 g; 89.1%). To a solution of indole (0.30 g, 2.6 mmol) in N,N-dimethylformamide (3 mL) was added 3,6-dibromo-1,4-dimethylpiperazine-2,5-dione (0.30 g, 1.0 mmol). The reaction mixture was stirred overnight, diluted with methanol and filtered to yield the bisindolyl product as a white, crystalline solid (0.34 g; 91.4%).
Author Comments
The reaction is an efficient way of adding indoles to the 3- and 6- positions of sarcosine anhydrides. A number of substituted indoles were utilized successfully. The 3,6-dibromo-1,4-dimethylpiperazine-2,5-dione is relatively unstable and should be reacted immediately.
Data
mp > 250°C; 1H NMR (d6-DMSO): 2.66 (s, 3H), 5.62 (s, 1H), 7.03 (t, 1H, J = 7.4), 7.13 (t, 1H, J = 7.4), 7.46 (m, 3H), 11.20 (s, 1H); MS: 372 (m+, 24.6), 255 (100.0), 227 (30.4), 159 (28.4), 157 (56.2).
Lead Reference
Whitlock, C. R. "Synthetic Approaches to the Dragmacidin Alkaloids." Dissertation, The University of Alabama, 1994.
Other References
Trown, P. W. Biochem. Biophys. Res. Commun. 1968, 33, 402.
Keywords
218, alkyl/alkenyl/aryl halides, amination, enzymatic, halogenation, insertion, organo phosphorous
Comments
Very interesting, did you use any special equipment to keep the Br2 from leaving the system?
By Vedran Hasimbegovic on February 8, 2006