Acylation of an alcohol
SyntheticPage 547
DOI:
Submitted: March 26, 2012, published: March 29, 2012
Authors
Rebecca Kaner (r.a.kaner@warwick.ac.uk)
A contribution from
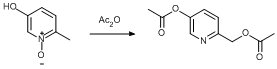
Chemicals
Acetic Anhydride
Procedure
5-Hydroxy-2-methylpyridine-1-oxide (22.33 g, 178.45 mmol) was suspended in acetic anhydride (250 ml). The clear yellow mixture was then stirred at reflux (150°C) for 3 h, causing a colour change to dark brown-black. The solution was cooled to ambient temperature and the solvent was removed under reduced pressure to leave a dark brown-black liquid, which was dried overnight in vacuo at 50°C. Yield = 36.59 g, 174.90 mmol, 98%.
Author Comments
Data
1H NMR (400 MHz, 298 K, DMSO) δH 8.39 (1H, d, 4JHH = 2.5 Hz, Py), 7.65 (1H, dd, 3JHH = 8.5 Hz, 4JHH = 2.5 Hz, Py), 7.49 (1H, d, 3JHH = 8.5 Hz, Py), 5.14 (2H, s, CH2), 2.31 (3H, s, CH3), 2.11 (3H, s, CH3).
13C{1H} NMR (100 MHz, 298K, DMSO) δC 170.0 (C=O), 169.0 (C=O), 152.9 (Py), 146.4 (Py), 142.7 (Py), 130.3 (Py), 122.4 (Py), 65.6 (CH2), 20.7 (CH3), 20.6 (CH3).
MS (ESI) m/z 210.1 [M+H]+, 232.1 [M+Na]+.
IR v cm-1 2946 w, 1765 m, 1738 s, 1581 w, 1485 m, 1436 w, 1370 m, 1182 s, 1024 s, 925 m, 898 m, 856 m, 722 w, 670 w.
Elemental Analysis found (Calculated for C10H11NO4) % C 57.46 (57.41), H 5.52 (5.30), N 6.63 (6.70).
Lead Reference
Supplementary Information
Keywords
acetic anhydride, esters, heterocyclic compounds