Esterification of Phosphorus Trichloride with Alcohols
SyntheticPage 488
DOI:
Submitted: March 18, 2011, published: March 20, 2011
Authors
Leandro Pedrosa (leandropedrosa@globo.com)
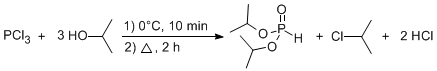
Chemicals
phosphorus trichloride 97%
isopropyl alcohol
Procedure
In a two-necked 125 mL round flask equipped with an addition funnel and a reflux condenser fitted with a bubbler, the dry isopropyl alcohol (0.70 mmol) was added. Through the addition funnel, freshly distilled PCl3 (0.23 mmol) was added dropwise with magnetic stirring so the temperature was kept around 0°C. After all the chloride was added, the mixture was kept under stirring at reflux for 2 h. The mixture was then kept under vacuum to remove residual HCl and finally the diisopropyl phosphonate was obtained a colorless liquid (36.6 g, 94%).
Author Comments
If the HCl were not completely removed, the diisopropyl phosphonate was liable to decompose.
The water can be removed from isopropyl alcohol by refluxing with CaO (200g/L) for several hours, then distilling.
The diisopropyl phosphonate can be purified by distillation obtained was 59°C - 0.2mmHg.
This particular reaction protocol can be applied to different alcohol.
Data
1H NMR (300 MHz, CDCl3) 1.31, 1.32 (12H, 2d, CH3, JHCCH = 5.9); 4.73 (2H, dhep, CH, JHCCH = 6.2, JPOCH = 8.5); 6.84 (1H, d, JPH = 688.28 Hz,)
13C NMR (75.42 MHz, CDCl3) 23.19 (d, CH3, JCCOP = 4.8); 23.37 (d, CH3, JCCOP = 4.3); 70.10 (d, CH, JCOP = 5.7)
31P NMR (121.42 MHz, CDCl3) 6.90
Lead Reference
McCombie, H.; Saunders, B. C.; Stacey, G. J.; J. Chem. Soc., 1945, 380. doi:10.1039/jr9450000380
Other References
Lin, W. O.; Souza, M. C.; DaCosta, J. B. N.; Quím. Nova, 1995, 18 (5), 431.
Fakhraian, H.; Mirzae, A.; Organic Process Research & Development 2004, 8, 401. doi:10.1021/op049958v
Rodrigues, J. M.; Sant'Anna, C. M. R.; Rumjanek, V. M.; DaCosta, J. B. N.; Phosphorus, Sulfur, and Silicon 2010, 185, 40. doi:10.1080/10426500903444366
Supplementary Information
Keywords
Esterification, organo phosphorous