Addition of oxirane to a thiol
SyntheticPage 446
DOI:
Submitted: July 25, 2010, published: July 27, 2010
Authors
Christopher Cooksey (rsc@chriscooksey.demon.co.uk)
A contribution from
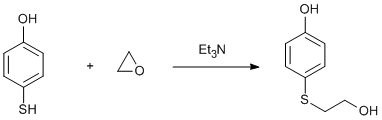
Chemicals
Ethylene oxide (Aldrich)
4-Mercaptophenol (Aldrich)
Triethylamine
Procedure
A mixture of hydroquinone (55 g, 0.5 mol), 1-bromopropane (24.6 g, 0.2 mol) was heated under N2 to reflux. A solution of KOH (11.2 g, 0.2 mol) in ethanol (80 cm3) was added over 20 m and heating continued for 4 h. After neutralisation (acetic acid, 20 cm3), evaporation gave a white solid which was extracted with chloroform (4 x 50 cm3). The chloroform was evaporated to give an oil containing white crystals (hydroquinone) which was dissolved in petroleum ether (60/80), filtering hot. After cooling, filtration gave white plates (9.72 g, 32%), m.p. 48-50 ºC. Ethylene oxide (3 cm3, 0.06 mol) was added to a test-tube equipped with a gas inlet and gas outlet and cooled in ice. A slow stream on N2 was passed through then into stirred 4-mercaptophenol (6.3 g, 0.05 mol) containing Et3N (3 drops) heated on a water bath at 45 – 50 ºC. After 1.5 h, all the ethylene oxide had gone but the reaction was incomplete (NMR) and more (2 x 1.5 cm3) was added, raising the temperature to 70 ºC since the product had solidified. The product was recrystallised from water – ethanol (7:1) to give slightly pink crystals (4.0 g, 47%), m.p. 72 – 74 ºC.
Author Comments
Lit: (Dachs et al) m.p. 71.8 – 73.0 ºC
Data
δH (DMSO-d6), 7.17d (2H), 6.65d (2H), 5.5brs (2H), 3.49t (2H), 2.80t (2H)
EI MS m/z(%), 170 (97), 140 (22), 139 (100)
mass 170.0410, calc for C8H12O2S 170.0402Lead Reference
Dachs NW, Gruber R, US patent, 1972, 3646220
Other References
Naish-Byfield S, Cooksey CJ, Latter AM, Johnson CI, Riley PA, Melanoma Res. 1991, 1(4), 273 – 287, PMID: 1823634
Keywords
electrophilic, epoxide, ethers, nucleophilic, ring-opening, sulphides