Preparation of (1,5-cyclooctadiene)chlororhodium (I) dimer
SyntheticPage 264
DOI:
Submitted: May 10, 2007, published: May 14, 2007
Authors
Kayla Collins (kayc_2@hotmail.com)
A contribution from
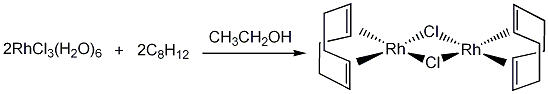
Chemicals
Rhodium trichloride hexahydrate (Precious Metals Online)
1,5-Cyclooctadiene (Aldrich)
1,5-Cyclooctadiene (Aldrich)
Procedure
The synthesis is performed under nitrogen, however, all work up procedures are carried out in air. A mixture of RhCl3(H2O)6 (2.0 g, 6.3 mmol), deoxygenated ethanol-water (5:1, 20 mL) and 1,5-cyclooctadiene (3 mL) were heated at reflux under nitrogen for 24 h, during this time period the product precipitated as a yellow-orange solid. After the reflux time was complete, the mixture was cooled to room temperature and the product was collected by filtration on a pre-weighed glass frit. The product was washed with pentane and methanol-water (v/v, 1:5, ~50 mL). The resulting yellow-orange powder was dried in vacuo (10-3 mbar) for 24 h. Yield: 1.47 g, 2.98 mmol, 95%.
Author Comments
During the washing procedure, the methanol-water filtrate is tested for the presence of the chloride ion using an aqueous silver nitrate solution. A “blank” solution of fresh methanol-water with a few drops of silver nitrate solution is used for comparison, once the washings achieve a similar level of clarity (low level of silver chloride precipitate), the washing procedure is stopped. In our experiences, there is no need to use sodium carbonate in this reaction, although it is used in the literature procedures. This Rh complex is a useful precursor for the synthesis of hydrogenation and hydroformylation catalysts. It can also be used to prepare cationic Rh precursors, including [Rh(COD)]BF4, by reaction with suitable silver salts.
Data
1H NMR (500 MHz, CDCl3) 1.75 (4 H, m, CH2), 2.52 (4H, m, CH2), 4.23 (4H, s, CH).
Lead Reference
G. Giordano and R. H. Crabtree, Inorganic Syntheses, 1979, 19, 218.
Other References
J. Chatt and L.M. Venanzi, J. Chem. Soc., 1957, 4735.