Sulfur transfer reaction of tetrathiomolybdate with a sulfonic ester
SyntheticPage 561
DOI:
Submitted: June 9, 2012, published: June 22, 2012
Authors
Ramesha Ramakrishna (ramesha63@hotmail.com)
A contribution from
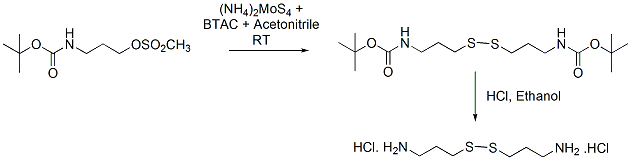
Chemicals
Ammonium tetrathiomolybdate, Aldrich
Benzyltriethylammonium chloride, commercial
Acetonitrile, Lab reagent
Ethyl acetate, Lab reagent
(N-tert-Butyloxycarbonyl)-3-aminopropyl mesylate -prepared by mesylating corresponding alcohol (Aldrich)
Ethyl alcohol
acetone
diethyl ether, lab grade
conc HCl
conc H2SO4
Benzyltriethylammonium chloride, commercial
Acetonitrile, Lab reagent
Ethyl acetate, Lab reagent
(N-tert-Butyloxycarbonyl)-3-aminopropyl mesylate -prepared by mesylating corresponding alcohol (Aldrich)
Ethyl alcohol
acetone
diethyl ether, lab grade
conc HCl
conc H2SO4
Procedure
Bis[(N-tert-butyloxycarbonyl)-3-aminopropyl]disulfide
Ammonium tetrathiomolybdate (1.1 g, 4.1 mmol) was added to a solution of benzyltriethylammonium chloride (2.1 g, 8.9 mmol) in acetonitrile (15 mL) and the mixture stirred for 1 h. The (N-tert-Butyloxycarbonyl)-3-aminopropyl mesylate (1 g, 4 mmol in 5 mL of acetonitrile) was added to the resultant red solution, and the mixture stirred for 18 h at room temperature. The reaction mixture was concentrated to approximately 50% of its original volume. Ethyl acetate (25 mL) was added to the dark mass, and the resultant slurry was filtered through Celite. Acetonitrile (5 mL) was added (to dissolve partly the remaining black residue in the flask), followed by ethyl acetate (25 mL), and the resultant slurry was filtered through the same Celite bed. Extraction of the solids was repeated as before, and the combined extracts were concentrated to dryness under reduced pressure. The residue was re-dissolved in ethyl acetate (100 mL), and the solution was washed with water to remove residual benzyltriethylammonium chloride. The ethylacetate layer was dried over sodiumsulfate and passed though a small path of silica and concentrated to get the desired disulfide as an oil (684 mg, 90% yield), that crystallizes on standing (mp: 82-83 oC).
Bis(3-aminopropyl)disulfide dihydrochloride
The above is(N-tertbutyloxycarbonyl)- 3-aminopropyl disulfide (0.68 g, 50 mmol) was dissolved in absolute ethyl alcohol (25 mL), and dry HCl gas (see below) was passed into the solution for 10 min while the temperature was maintained below 50-60 oC. On concentration of the ethanol, the dihydrochloride precipitates. The white solid was triturated with ether, filtered, dried, and stored in a vacuum desiccator (0.44 g, 97% yield).
Generation of dry hydrogen chloride: 200 mL concentrated HCl (about 30-35%) charged to a clean 1 l flask carrying a 250 mL additional flask. Top of the addition flask is connected to a glass bubble trap containing 20 mL concentrated sulphuric acid (which acts as drying agent for HCl gas). About 200 mL concentrated sulphuric acid is charged to the addition flask. Drop wise addition of sulphuric acid instantaneously generates HCl gas, which passes through sulphuric acid bubble trap and comes out as dry HCl gas. This can be directly purged to the reaction mixture by a glass tube. This method is superior to sodium chloride+sulphuric acid method as the generation is more uniform. Almost 90-95% of theoretical (based on the HCl used) HCl gas is liberated.
Ammonium tetrathiomolybdate (1.1 g, 4.1 mmol) was added to a solution of benzyltriethylammonium chloride (2.1 g, 8.9 mmol) in acetonitrile (15 mL) and the mixture stirred for 1 h. The (N-tert-Butyloxycarbonyl)-3-aminopropyl mesylate (1 g, 4 mmol in 5 mL of acetonitrile) was added to the resultant red solution, and the mixture stirred for 18 h at room temperature. The reaction mixture was concentrated to approximately 50% of its original volume. Ethyl acetate (25 mL) was added to the dark mass, and the resultant slurry was filtered through Celite. Acetonitrile (5 mL) was added (to dissolve partly the remaining black residue in the flask), followed by ethyl acetate (25 mL), and the resultant slurry was filtered through the same Celite bed. Extraction of the solids was repeated as before, and the combined extracts were concentrated to dryness under reduced pressure. The residue was re-dissolved in ethyl acetate (100 mL), and the solution was washed with water to remove residual benzyltriethylammonium chloride. The ethylacetate layer was dried over sodiumsulfate and passed though a small path of silica and concentrated to get the desired disulfide as an oil (684 mg, 90% yield), that crystallizes on standing (mp: 82-83 oC).
Bis(3-aminopropyl)disulfide dihydrochloride
The above is(N-tertbutyloxycarbonyl)- 3-aminopropyl disulfide (0.68 g, 50 mmol) was dissolved in absolute ethyl alcohol (25 mL), and dry HCl gas (see below) was passed into the solution for 10 min while the temperature was maintained below 50-60 oC. On concentration of the ethanol, the dihydrochloride precipitates. The white solid was triturated with ether, filtered, dried, and stored in a vacuum desiccator (0.44 g, 97% yield).
Generation of dry hydrogen chloride: 200 mL concentrated HCl (about 30-35%) charged to a clean 1 l flask carrying a 250 mL additional flask. Top of the addition flask is connected to a glass bubble trap containing 20 mL concentrated sulphuric acid (which acts as drying agent for HCl gas). About 200 mL concentrated sulphuric acid is charged to the addition flask. Drop wise addition of sulphuric acid instantaneously generates HCl gas, which passes through sulphuric acid bubble trap and comes out as dry HCl gas. This can be directly purged to the reaction mixture by a glass tube. This method is superior to sodium chloride+sulphuric acid method as the generation is more uniform. Almost 90-95% of theoretical (based on the HCl used) HCl gas is liberated.
Author Comments
1. These diaminodisulfides are used in crosslinking DNA and are difficult to prepare from conventional procedures.
2. This procedure gives almost quantitative yield in pure form.
3. During disulfide formation, yield depends on how effectively the product is extracted from the molybdenum sludge.
4. No aqueous workup is done. The reaction is done under almost neutral condition.
5. This reaction also works if the leaving group is chloro or bromo or tosylate.
2. This procedure gives almost quantitative yield in pure form.
3. During disulfide formation, yield depends on how effectively the product is extracted from the molybdenum sludge.
4. No aqueous workup is done. The reaction is done under almost neutral condition.
5. This reaction also works if the leaving group is chloro or bromo or tosylate.
Data
Bis[(N-tert-butyloxycarbonyl)-3-aminopropyl]disulfide: 1H NMR (300 MHz, CDCl3) d 4.68 (s, br, 2H), 3.24 (m, 4H), 2.72 (t, 4H), 1.88 (m, 4H), 1.45 (s, 18H). MS (FAB) m/z: 381 (M+1), 325, 225, 102, 90. HRMS m/z, calculated for C16H32O4N2S2, 380.1804; found, 380.1882.
Bis(3-aminopropyl)disulfide dihydrochloride : 1H NMR (300 MHz, CD3OD) d 3.10 (t, 4H), 2.80 (t, 4H), 2.10 (q, 4H).
Bis(3-aminopropyl)disulfide dihydrochloride : 1H NMR (300 MHz, CD3OD) d 3.10 (t, 4H), 2.80 (t, 4H), 2.10 (q, 4H).
Lead Reference
Modified procedure of
1. A. R. Ramesha, Chandrasekaran S., Synthetic Communications, 1992, 22, 3277–3284.
2. Stefan G. Sarafianos, Arthur D. Clark Jr., Steve Tuske, Chris J. Squire, Kalyan Das, Dequan Sheng, Palanichamy Ilankumaran, Andagar R. Ramesha, Heiko Kroth, Jane M. Sayer, Donald M. Jerina, Paul L. Boyer, Stephen H. Hughes, and Eddy Arnold. The Journal of Biological Chemistry, 2003, Vol. 278, Issue 18, 16280-16288
Keywords
Alkanes, amines, diamine, disulfide, esters, hydrochloride, nucleophilic, substitution, sulfur, sulphides