Nucleophilic aromatic substitution
SyntheticPage 556
DOI:
Submitted: May 2, 2012, published: May 7, 2012
Authors
Alex Henderson (Alex.Henderson@bris.ac.uk)
A contribution from
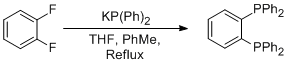
Chemicals
Potassium diphenylphosphide solution, 0.5 M in THF (Sigma-Aldrich)
1,2-Difluorobenzene, 98% (Sigma-Aldrich)
PhMe (Grubbs Solvent System - passed through passed column of alumina)
Activated charcoal (Sigma-Aldrich)
1,2-Difluorobenzene, 98% (Sigma-Aldrich)
PhMe (Grubbs Solvent System - passed through passed column of alumina)
Activated charcoal (Sigma-Aldrich)
Procedure
To an anhydrous 0.5 M THF solution of potassium diphenylphosphide (45.0 mL, 22.5 mmol) at RT, was added 1,2-difluorobenzene (1.97 mL, 20.0 mmol) and anhydrous N2 saturated PhMe (50.0 mL). The resulting solution was heated to reflux (110 °C) for 5 h, by which time the dark red colour of the phosphide had dissipated, and 19F NMR spectroscopy showed a single environment (δF: -103.3, d, JFP = 53.0 Hz) correlating to a monophosphine species.§ To the solution at RT, was added an anhydrous 0.5 M THF solution of potassium diphenylphosphide (45.0 mL, 22.5 mmol) and left to reflux (110 °C) for 15 h. P NMR spectroscopy revealed a single peak (δp: -12.3) and the red/orange reaction mixture was concentrated in vacuo. The residue was suspended in PhMe (200 mL) at 70 °C, and activated charcoal (≈ 5.0 g) was added, whilst vigorous stirring occurred. The mixture was hot vacuum filtered through Celite and the solvent was removed in vacuo. The crude material was recrystallised (PhMe) to afford 1,2-bis(diphenylphosphino)benzene (87%, 7.80 g) as a colourless solid.
§No peak observed for 1,2-difluorobenzene (δF: -138.7). P NMR spectrum showed concurrent data (δP: -18.0, d, JPF = 53.0 Hz).
§No peak observed for 1,2-difluorobenzene (δF: -138.7). P NMR spectrum showed concurrent data (δP: -18.0, d, JPF = 53.0 Hz).
Author Comments
- Glassware was oven dried (150 oC) prior to use.
- Reaction performed under an atmosphere of N2 in purged apparatus.
- NMR spectroscopy was carried out with aliquots from the reaction vessel (taken in an inert fashion) and later recombined.
- All chemicals were used without further purification however were checked for purity by 1H and 31P NMR spectroscopy.
- KP(Ph)2 was transferred to a Young's flask, from the Sigma-Aldrich bottle, prior to use.
- Celite filter additionally washed with hot (70 oC) PhMe (2 x 40 mL) as product precipitates in cool PhMe.
- After recrystallisation, the solution was naturally left to cool to RT which caused precipitation of some material. The RBF was then refridgerated for 4 h which enhanced precipitation. Compound isolated by Buchner filtration.
- Product is air stable.
Data
νmax / cm-1 (film): 3056 (w), 1583 (w), 1567 (w), 1476 (m), 1435 (m), 1309 (w), 1157 (w), 1069 (w); mp 186-187 °C (PhMe) [Lit. 183-185 °C (PhMe / EtOH)]; δH (400 MHz, CDCl3): 6.97-7.02 (4H, m, ArCH), 7.08-7.22 (20H, m, ArCH); δC(100 MHz, CDCl3): 128.3 (t, J = 3.5 Hz, ArCH), 128.4 (ArCH), 129.1 (ArCH), 133.9 (t, J = 10.0 Hz, ArCH), 134.1 (t, J = 3.0 Hz, ArCH), 137.1 (t, J = 3.0 Hz, ArC), 143.7 (dd, J = 10.0 and 10.0 Hz, ArC); δp (121 MHz, CDCl3): -13.4.
Lead Reference
Baker, B. A.; Bošković, Ž. V.; Lipshutz, B. H. Org. Lett. 2008, 10, 289-292.
Other References
McFarlane, H. C. E.; McFarlane, W. Polyhedron. 1983, 2, 303-304.
Tunney, S. E.; Stille, J. K. J. Org. Chem. 1987, 52, 748-753.
Tunney, S. E.; Stille, J. K. J. Org. Chem. 1987, 52, 748-753.
Keywords
aromatic nucleophilic substitution, aromatics/arenes, carbocyclic compounds, ligand, organo phosphorous