iodoalkynes from aldehydes
SyntheticPage 24
DOI:
Submitted: July 3, 2001, published: July 3, 2001
Authors
zac etheridge (zac.etheridge@orange.net)
A contribution from
Chemicals
Triphenylphosphine (1.15g, 4mmol)
Iodoform (1.65g, 4.2mmol)
Potassiumtbutoxide (1.569g, 12mmol)
Cinnamaldehyde (0.252mL, 2mmol)
THF (dry, 20mL)
Iodoform (1.65g, 4.2mmol)
Potassiumtbutoxide (1.569g, 12mmol)
Cinnamaldehyde (0.252mL, 2mmol)
THF (dry, 20mL)
Procedure
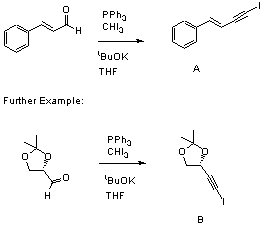
To iodoform stirring in THF is added triphenylphosphine and 1 equivalent of potassiumtbutoxide and the brown mixture is stirred for 1 minute. Cinnamaldehyde is added and the reaction stirred for 15 minutes, then cooled to -78C. The remaining potassiumtbutoxide is then added and the reaction stirred until completion (TLC immediately after the second addition of potassiumtbutoxide in order to ascertain which spot is the intermediate) then quenched with brine, extracted twice with ether, dried over magnesium sulphate, filtered, concentrated and purified by colummn chromatography eluting with 10% Et2O/PE.
Author Comments
This procedure works extremely well and has been carried out on scales of upto 20g using the glyceraldehyde derivative (B) shown in the illustration. Note that in this instance, the aldehyde is more unstable and it's additon should be carried out at 0C. Yields of 90-95% are attainable, although in the case of the glyceraldehyde derivative, the yield is lower and highly dependant on the method used to make the aldehyde. The reaction proceeds via a diiodoalkene, which can easily be isolated by stopping the reaction before adding the second batch of potassiumtbutoxide.
Data
A:13CNMR (CDCl3, 75MHz) 8.1, 93.4, 108.1, 126.3, 128.8, 128.9, 135.4, 143.4. B:1HNMR (CDCl3, 300mHz) d 4.71 (1H, t, J=6.2), 4.03 (1H, dd, J=6.4, 8.1), 3.82 (1H, dd, J=6.2, 8.1), 1.37 (3H, s), 1.25 (3H, s).
Lead Reference
Michel P., Rassat A. Tetrahedron Lett. 1999 p.8579
Other References
Hirama M., Iguchi S., Wang G.X. J. Org. Chem. 2001 p.2146
Keywords
iodoalkyne, alkyne, diiodoalkene, aldehyde, ylid, Speziale, Wittig, isotopically labelled compounds